Atomic orbitals2/20/2023  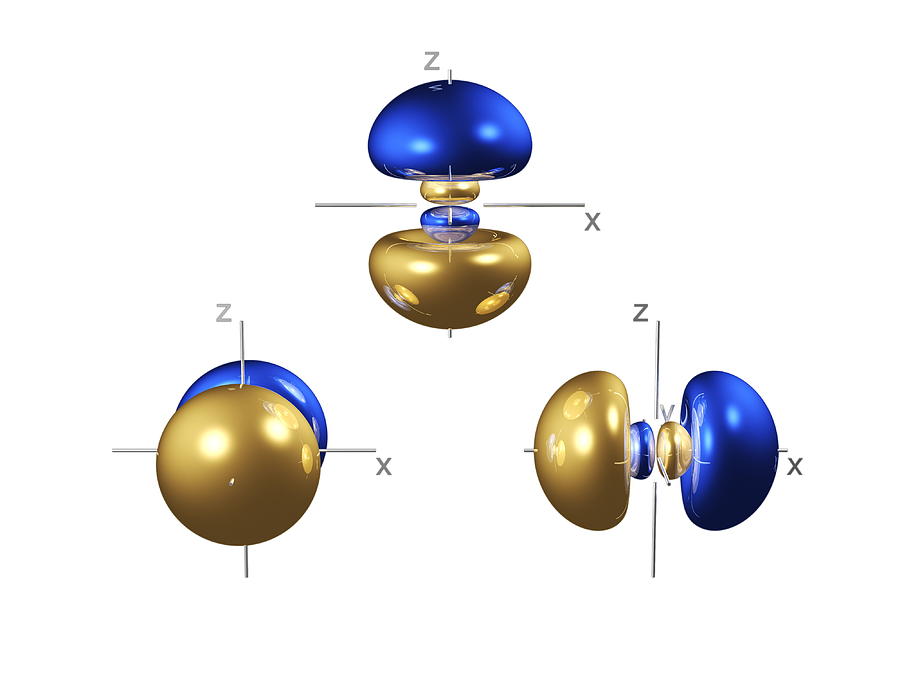
When four atomic orbitals are added together, four hybrid orbitals form. Hybridization is an internal linear combination of atomic orbitals, in which the wave functions of the atomic s and p orbitals are added together to generate new hybrid wave functions. The creation of identical orbitals occurs in nature by a hybridization process. To achieve equal bond lengths, all the orbitals would have to be the same type. However, these bonds would not all be of the same length because atomic 5 orbitals are shorter than atomic p orbitals. The excited state has four half‐filled orbitals, each capable of forming a covalent bond. Adding energy to the system promotes a 2 s electron to a 2 p orbital, with the resulting generation of an excited state. The ground state, or unexcited state, of the carbon atom ( Z = 6) has the following electron configuration.Ĭovalent bonds are formed by the sharing of electrons, so ground‐state carbon cannot bond because it has only two half‐filled orbitals available for bond formation. all of the bond angles are approximately 110°.all of the hydrogen‐carbon‐hydrogen bond angles are equal.all of the carbon‐hydrogen bond lengths are equal.Physical studies of the simplest organic compound, methane (CH 4), have shown the following: Cahn‐Ingold‐Prelog RS Notational System.Racemic Mixtures: Resolving Enantiomers.Cyclohydrocarbons: Reactivity, Stresses of Small Rings.Alkynes: Molecular and Structural Formulas. 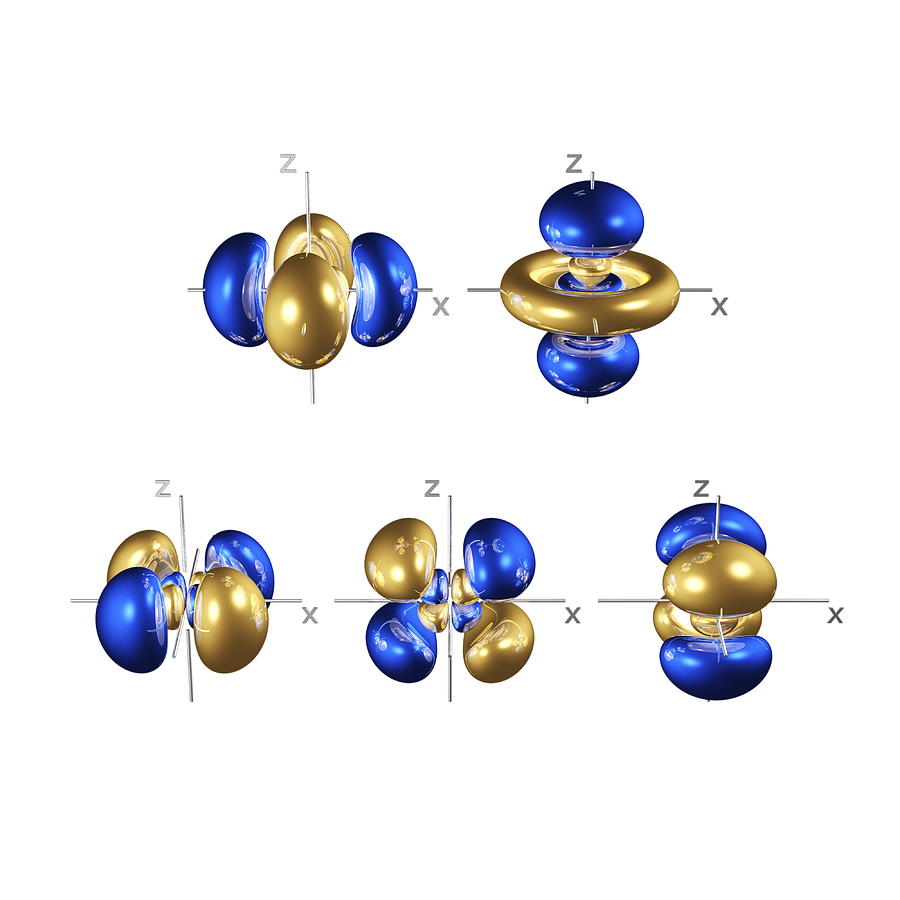
Alkenes: Oxidation and Cleavage Reactions.Alkenes: Electrophilic Addition Reactions.Alkenes: Catalytic Addition of Hydrogen.Alkenes: Hydration (Direct Addition of Water). 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
